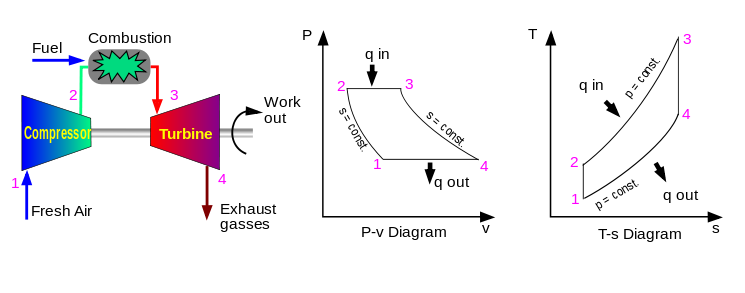
Thermodynamic is a #science of the relationship between heat ( thermo ) , work (dynamics) .

A thermodynamic cycle is a series of thermodynamic processes which returns a system to its initial state
Here are the most famous thermodynamic cycles :
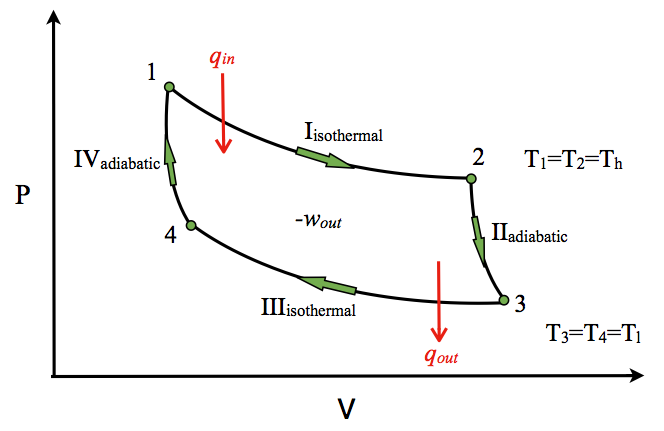
1- #Carnot Cycle :
The Carnot cycle consists of the following four processes:
- A reversible isothermal gas expansion process. In this process, the ideal gas in the system absorbs qin amount heat from a heat source at a high temperature Th, expands and does work on surroundings.
- A reversible adiabatic gas expansion process. In this process, the system is thermally insulated. The gas continues to expand and do work on surroundings, which causes the system to cool to a lower temperature, Tl.
- A reversible isothermal gas compression process. In this process, surroundings do work to the gas at Tl, and causes a loss of heat, qout.
- A reversible adiabatic gas compression process. In this process, the system is thermally insulated. Surroundings continue to do work to the gas, which causes the temperature to rise back to Th.
carnot thermodynamic cycle on p-v chart It used in refrigeration or as a thermal pump.
2- #Rankine cycle :The Rankine cycle is an idealized thermodynamic cycle of a heat engine that converts heat into mechanical work while undergoing phase change as water evaporation and steam condensing ..
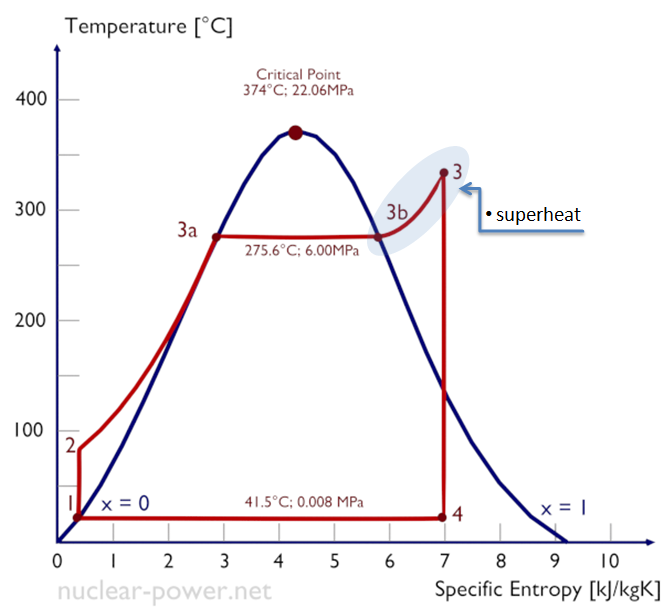
it used in #steam #turbine to convert heat energy into work in power plants or steam #engine .
3- #Otto cycle : The Otto cycle is a set of processes used by spark ignition internal combustion engines
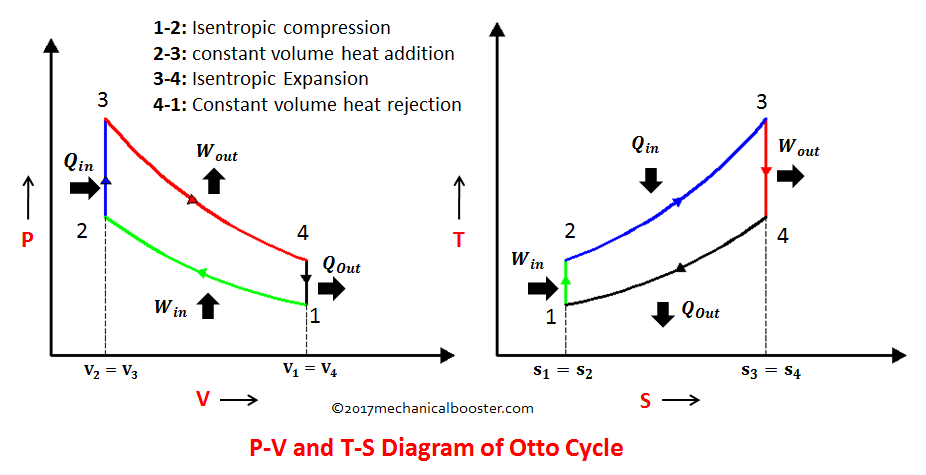
1. Isentropic ( reversible adiabatic) compression
2. Constant volume (Isochoric) heat addition
3. Isentropic (reversible adiabatic) Expansion
4. Constant volume heat rejection.
4- #Diesel cycle : Diesel cycle is widely used in diesel engines. Diesel cycle is similar to Otto cycle except in the fact that it has one constant pressure process instead of a constant volume process (in Otto cycle).

5- #Brayton cycle : The Brayton cycle is the ideal cycle for gas-turbine engines.